
What Are Some Transition Metals. Sometimes included in the transition metal group are the lanthanides and actinides. They include iron copper and chromium. There are a number of elements that are classified as transition metals. A transition metal is one that forms one or more stable ions which have incompletely filled d orbitals.
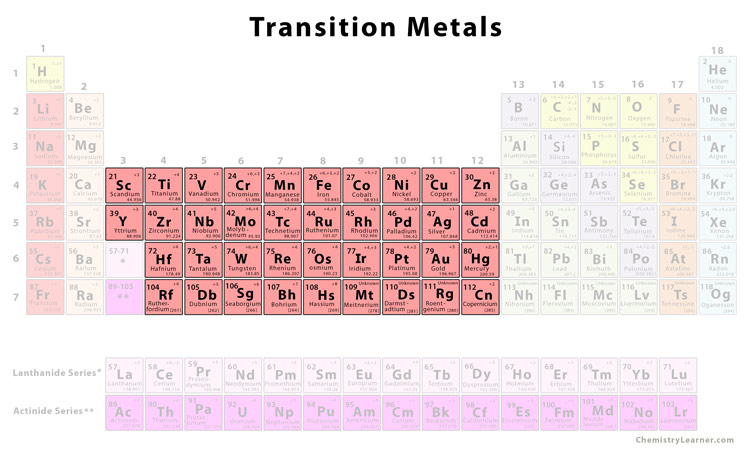
List of Elements That Are Transition Metals Scandium Titanium Vanadium Chromium Manganese Iron Cobalt Nickel Copper Zinc Yttrium Zirconium Niobium Molybdenum Technetium Ruthenium Rhodium Palladium Silver Cadmium Lanthanum sometimes often considered a rare earth lanthanide Hafnium Tantalum. Diamagnetic compounds have d-electrons that are all paired up. Sometimes included in the transition metal group are the lanthanides and actinides. They occupy columns 3 through 12 of the periodic table and include such metals as titanium copper nickel silver platinum and gold. Transition metal any of various chemical elements that have valence electronsie electrons that can participate in the formation of chemical bondsin two shells instead of only one. Some of these metallic elements include chromium copper cobalt iron mercury manganese tin lead and bismuth.
Typically transition elements are precious and elements with special characteristics in the periodic table.
There are a total of 38 elements which are known as Transition Metals. Together they make up the largest section of the periodic table as it includes metals like copper gold and titanium. Transition elements are the elements which are in the groups 3 to 12 or III B to VIII. Besides that transition elements located in between group 2 and group 13 in the periodic table each series consist of 10 elements. Transition metals do however form a very wide range of complex ions. Transition metals are like main group metals in many ways.