Why Is Infrared Spectroscopy Sometimes Referred To As Vibrational Spectroscopy. Infrared spectroscopy IR spectroscopy or vibrational spectroscopy involves the interaction of infrared radiation with inorganic chemicals and covers a range of techniques mostly based on absorption spectroscopy. As with all spectroscopic techniques it can be used to identify and study inorganic chemicals. For a given sample which may be solid liquid or gaseous the method or technique of. It covers a range of techniques mostly based on absorption spectroscopy.
For a given sample which may be solid liquid or gaseous the method or technique of. The main use of this technique is in organic and inorganic chemistry. This covers infrared near-infrared and Raman spectroscopies and publishes papers dealing with developments in applications theory techniques and instrumentation. A collisional model provides similar insight in a gas and a non-H. Chemical compounds have different chemical properties due to the presence of different functional groups. IR Spectroscopy measures the vibrations of atoms and based on this it is possible to.
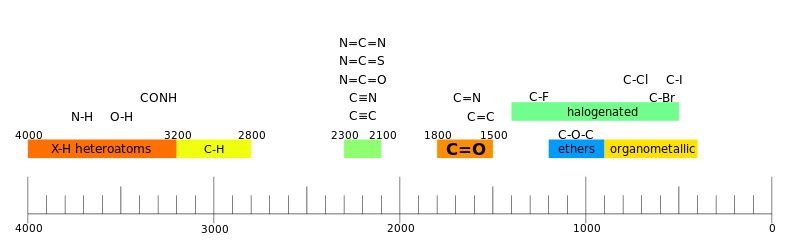
In infrared spectroscopy the sample is irradiated with polychromatic light and a photon of light is absorbed when the frequency energy of the absorbed light matches the energy required for a particular bond to vibrate within the sample.
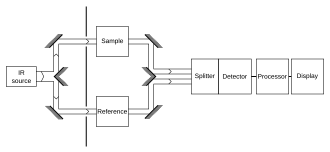
Vibrational Spectroscopy IR Raman Vibrational spectroscopy. The main use of this technique is in organic and inorganic chemistry. Infrared IR spectroscopy or vibrational spectroscopy is an analytical technique that takes advantage of the vibrational transitions of a molecule. A common laboratory instrument that uses. For all those reason IR called Vibrational spectra. In order for a vibration to be infrared active the molecular dipole moment must change during the vibration.